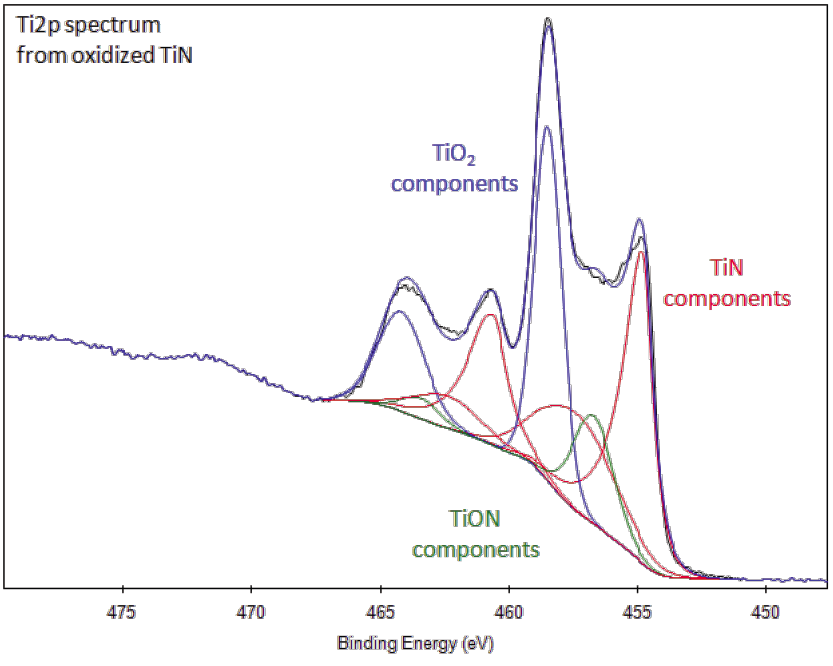
Titanium is not found unbound in nature and it occurs mainly in minerals. In quantitative XPS analysis problems arise due to difficulties in defininq Ti 2p peak areas.
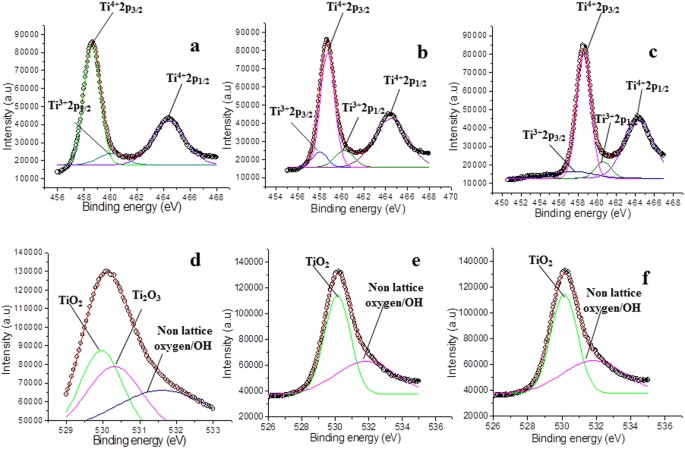
Because titanium is physiologically inert, it is used in joint replacement implants and is popular in body piercing. because of the overlap of titanium and nitrogen Auger peaks. AES shows a strong overlapping of the most intensive peaks of Ti and N. Paint made with TiO2 reflects infrared radiation, making it useful in sunscreen and to astronomers. induced Photoelectron Spectroscopy (XPS) data. Its silvery metallic color makes it very useful in paints, paper and plastics. Most titanium is used in the form of titanium dioxide (TiO2). Titanium has a very high strength-to-weight ratio and is corrosion-resistant. Sub-oxides and/or metal may be formed during sputtering of the oxide.TiO 2 is readily reduced by argon monomer sputtering.Titanium is used as a getter material for oxygen.Unleash the power to create: With Intel ® ’s latest 10th gen, 8 core, 16 threads i7 processor, you can keep at your most demanding workloads for even longer. High-resolution component peak fits of, a) Ti 2p, b) C 1s, c) O 1s, and d) F 1s regions. Causes difficulty in accurately peak fitting Ti2p region with multiple chemical The XPS 15: Dell Creator Edition is fully equipped with the creative tools you need to make magic happen faster and better, so your art can be pushed to the edge. Figure 1: Component peak fits of XPS spectra of as-prepared Ti 3 C 2 T z powders.(Post-ionization, Ti2p 1/2 state is very short lived Typically FWHM for each spin-orbit component is the same, but for Ti2p the Ti2p 1/2Ĭomponent is much broader than the Ti2p 3/2 peak.Ĭonsequently, Ti2p 1/2 peak is much shorter than expected.Splitting -value varies with chemical state (Δ nitride=6.0eV, Δ oxide=5.7eV).Ti2p peak has significantly split spin-orbit components (Δ metal=6.1eV).O TiO2 has symmetric peaks shapes and TiN has a complex peak shape, involving satellite

Use XPS valence band spectrum to distinguish between these oxides. SnO and SnO 2 have similar binding energies. Loss features are observed to higher binding energy side of 3d 3/2 spin-orbit component for Sn metal. Keywords: TiN, XPS, magnetron sputtering, binding energy, VN, CrN, NbN. Peaks have asymmetric peak shape for metal. Ti metal gives asymmetric Ti2p peaks shapes High energy resolution core level XPS spectra acquired with monochromatic Al.Binding energies of common chemical states:
